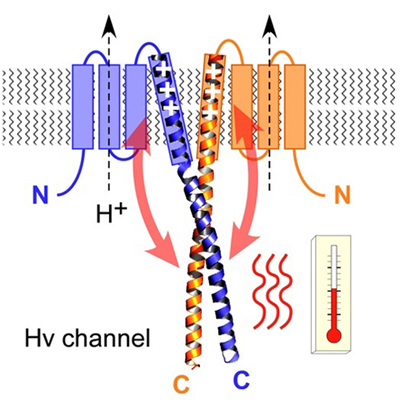
Hv1/VSOP is a dimeric voltage-gated H+ channel in which the gating of one subunit is reportedly coupled to that of the other subunit within the dimer. The molecular basis for the dimer formation and intersubunit coupling remains unknown, however. Here we show that the C-terminus ends downstream of the S4 voltage sensor helix twist into a dimer coiled-coil architecture which mediates the cooperative gating. We also show that the temperature-dependent activation of H+current through Hv1 is regulated by the thermostability of the coiled-coil domain, and the regulation is altered by mutation into the linker between S4 and the coiled-coil. The cooperative gating within the dimer is also dependent on the linker structure which is likely α-helical as suggested by CD spectrum analysis. Our results indicate that the cytoplasmic coiled-coil strands forming continuous α-helices with S4 mediate the cooperative gating and adjust the range of temperature over which Hv1 operates.
(Fujiwara et. al., Nature Communications 3: 816, 2012)
*Integrative Physiology, Department of Physiology, Graduate School and Faculty of Medicine, Osaka University