Physiological body temperature is an important determinant for neural functions, and it is well established that changes in temperature have dynamic influences on hippocampal neural activities. However, the detailed molecular mechanisms have never been clarified. Here, we show that hippocampal neurons express functional TRPV4, one of the thermo-sensitive transient receptor potential (TRP) channels, and that TRPV4 is constitutively active at physiological temperature. Activation of TRPV4 at 37°C depolarized the resting membrane potential (RMP) in hippocampal neurons by allowing cation influx, which was observed in wild-type (WT) neurons, but not in TRPV4-deficient (TRPV4KO) cells, although dendritic morphology, synaptic marker clustering and synaptic currents were indistinguishable between the two genotypes. Furthermore, current injection studies revealed that TRPV4KO neurons required larger depolarization to evoke firing, equivalent to WT neurons, indicating that TRPV4 is a key regulator for hippocampal neural excitabilities. We conclude that TRPV4 is activated by physiological temperature in hippocampal neurons and thereby controls their excitability.
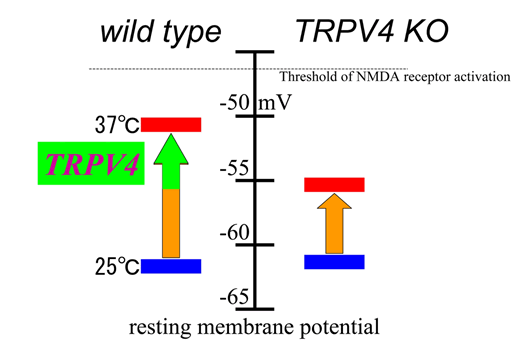
Fig. A proposed model for TRPV4 involvement in the regulation of resting membrane potentials (RMP) in hippocampal pyramidal neurons.
RMP were not significantly different between wild-type and TRPV4KO neurons at ~ –62 mV at 25oC. RMP were significantly depolarized to a value close to the threshold for NMDA receptor activation in wild-type neurons compared with TRPV4KO at 37oC, leading to more excitability.
*Section of Cell Signaling, Okazaki Institute for Integrative Bioscience